Hip Implants With the Potential to Be Long Lasting
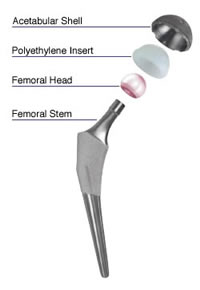
The durability of hip implants depends on many things including patient weight and activity level, as well as the implant’s bearing surface technology.
Improved Wear Performance
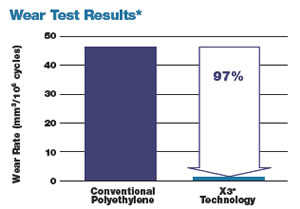
The bearing surface is defined as the two parts of the hip that glide together throughout motion. Stryker’s new, advanced bearing technology, called X3®, has demonstrated up to 97% decrease in wear in laboratory testing.* This decrease in wear may extend the life of your hip implant. This new technology is particularly important for younger patients.
Ask your doctor if X3® technology is right for you.
Stryker Corporation or its divisions or other corporate affiliated entities own, use or have applied for the following trademarks or service marks: Stryker and X3. All other trademarks are trademarks of their respective owners or holders.
References:
Stryker® Orthopaedics Trident® Acetabular Inserts made of X3® UHMWPE (unsterilized), 721-00-32E, show a 97% reduction in volumetric wear rate versus the same insert fabricated from N2\Vac™ gamma sterilized UHMWPE, 620-00-32E.The insert tested was 7.5mm thick with an inner diameter of 32mm. Testing was conducted under multi-axial hip joint simulation for 5 million cycles using a 32mm CoCr articulating counterface and calf serum lubricant. X3® UHMWPE Trident® Acetabular Inserts showed a net weight gain due to fluid absorption phenomena but yielded a positive slope and wear rate in linear regression analysis. Volumetric wear rates were 46.39 ± 11.42mm3/106 cycles for N2\Vac™ gamma sterilized UHMWPE inserts and 1.35 ± 0.68mm3/106 cycles for X3® UHMWPE (unsterilized) Trident® Acetabular Inserts. Although in-vitro hip wear simulation methods have not been shown to quantitatively predict clinical wear performance, the current model has been able to reproduce correct wear resistance rankings for some materials with documented clinical results.a, b, c
a. Wang, A., et al., Tribology International, Vol. 31, No. 1-3:17-33, 1998.
b. Essner, A., et al., 44th Annual Meeting, ORS, New Orleans,Mar. 16-19, 1998:774.
c. Essner, A., et al., 47th Annual Meeting, ORS, San Francisco, Feb. 25-28, 2001:1007.
* Stryker Orthopaedics Test Report: RD-03-082.

